 The study was performed regarding the influence of pH, time of contact, dosage of sorbent, initial metal ions concentration and temperature. The present study was performed to compare the potential removal of Cr 3+ and Cr 6+ using mango peels from aqueous solution. Recently, different parts of the mango such as mango seeds, mango peels, unfertilizable fruiting buds of mango plant and phosphate treated sawdust of mango tree have been employed to sequester dyes, phenol, heavy metal ions. Due to these functional groups, the mango peels sequester different types of pollutants, especially metal ions. Cellulose and pectin contain various functional groups such as carboxylic and hydroxyl. The major components of mango peels are pectin, lipids, cellulose, hemicellulose, polyphenols and carotenoids. These peels have no proper use and the disposal of these wastes is also a serious problem. About 7-14% of the total fruit weight is the peels of mango and generated in a huge amount during the season. The edible part of the mango is the pulp making approximately 33 to 85% of the total fruit and used in many products include juices, jams and jelly powders. The annual production of the mango is about 27 million tons which makes the mango on fifth position among major fruits. Mango is one of the delicious fruit and commonly grows in those regions of the world which occur on tropical and subtropical. It has been reported that those materials which are obtained from various peels of the fruit have a high potential to remove heavy metal ions. On comparison to other conventional wastewater treatment technologies, biosorption has many potential marketing advantages such as high efficiency, low cost, no additional nutrient requirement, low biological sledge formation, eco-friendly, recycling of biosorbents, high metal binding ability and high possible metal recovery. Recently, biosorption has become one of the alternative technologies that have been widely utilized for the decontamination of wastewater. Therefore, it is imperative to investigate an inexpensive and eco-friendly method to remove chromium and other contaminants from water. However, there are a number of considerable drawbacks of these methods such as high price, energy consumption, partial removal and further production of toxic sledge due to which they have not gained much attention. Various methods have been applied so far, to treat polluted water like ion exchange, precipitation, solvent extraction, electrochemical deposition, membrane separation, adsorption, reduction etc. Hence, the removal of chromium from the industrial effluents is necessary before discharging into the environment. However, maximum exposure of the chromium causes serious health problems such as cancer, allergic skin reaction, kidney and lung diseases. 
The literature showed that Cr 3+ is an important nutrient necessary for the metabolism of sugar and fat, and its toxicity is less than Cr 6+. Among these metals, chromium is considered as toxic and usually found in the form of Cr 3+ and Cr 6+. 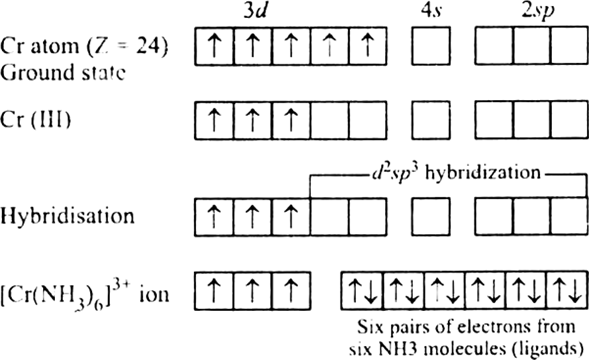
The effluents of various industries such as electroplating, tanning, metallurgy, pesticide, ceramic, batteries and mining consist of heavy metals in high amount. 
The accumulation of these metals in the bodies of flora and fauna causes serious problems. Scientists have focused their attention much more on the contamination of heavy metals. The spontaneity and exothermic nature of the sorption process of Cr 3+ and Cr 6+ using mango peels was reflected from thermodynamic study. 
The monolayer sorption capacity of mango peels was found to be 98.039 mg g -1 for Cr 3+ and 66.666 mg g -1 for Cr 6+. The isotherms study revealed that Freundlich is the suitable isotherm for explanation of the equilibrium data due to higher R 2 values. Among various kinetic models, pseudo-2 nd order well explained the data owing to the higher values of R 2 and the nearness between the values of experimental and calculated sorption capacities. The pH study revealed that that maximum removal of Cr 3+ and Cr 6+ was obtained at pH 5.0 and 7.0 respectively. Characterization of the mango peels was done before and after sorption of Cr 3+ and Cr 6+ using scanning electron microscopy, surface area pore size analyzer and FTIR spectroscopy. The present study investigates a comparative study of the sorption of Cr 3+ and Cr 6+ from water using an agricultural by-product mango peels in batch system under the effect of initial metal ion concentrations, solution pH, temperature, sorbent dose and contact time. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
